Moderna Booster Authorization
The company submitted data to the FDA on Wednesday. Food and Drug Administration FDA to allow the use of a third booster dose of its COVID-19 vaccine.

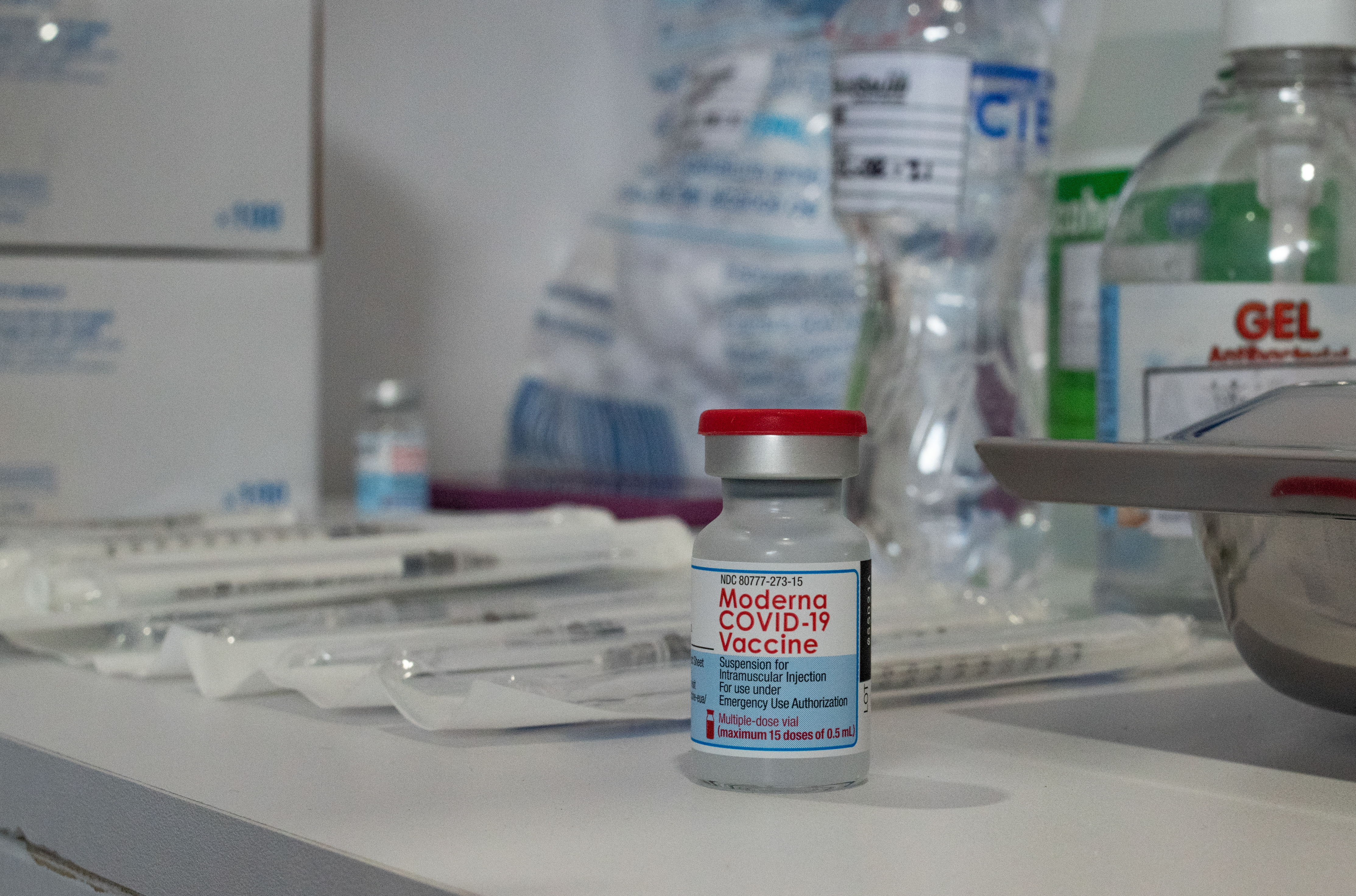
Moderna Vaccine Nbc 7 San Diego
The company also said it had completed.
Moderna booster authorization. In addition to Moderna potentially adding a half-dose booster Pfizer is seeking authorization of a vaccine for children ages 5 to 11 with a 10-microgram dose -- one-third the strength given to. The FDA is considering booster shots of the Pfizer and BioNTech vaccine but so far has only allowed people with weakened immune systems to receive third doses of either the Moderna or Pfizer shots. About 170 million fully vaccinated people in the US.
The company also said it had completed. In addition to Moderna potentially adding a half-dose booster Pfizer is seeking authorization of a vaccine for children ages 5 to 11 with a 10. Moderna like Pfizer is hoping to gain authorization for its vaccine booster shot ahead of the September 20 roll out date.
Food and Drug Administration FDA to allow the use of a third booster dose of its COVID-19 vaccine. Moderna on Wednesday asked the US. The authorization would set the stage to further widen the US.
Reuters -Moderna Inc on Wednesday asked the US. Moderna seeks US. The agency said on Wednesday that a.
The FDA is considering booster shots of the Pfizer and BioNTech vaccine but so far has only allowed people with weakened immune systems to receive third doses of either the Moderna or Pfizer shots. Moderna MRNA is developing a booster dose of its COVID-19 vaccine mRNA-1273 for enhancing protection against infection in individuals. Moderna is seeking FDA authorization for a third dose of its COVID-19 vaccine for the general public.
1 2021-- Moderna Inc. Authorization for COVID-19 vaccine booster. Food and Drug Administration FDA to allow the use of a third booster dose of its COVID-19 vaccine.
The FDA is considering booster shots. The filing adds fuel to a debate about whether COVID booster shots are needed. Christina Rostad an assistant professor of.
Food and Drug Administration FDA to allow the use of a third booster dose of its COVID-19 vaccine. Moderna Inc on Wednesday asked the US. Moderna on Sept.
Moderna awaits FDA decisions on full approval for its COVID-19 vaccine and for booster doses. WASHINGTON REUTERS - Moderna on Wednesday Sept 1 asked the United States Food and Drug Administration FDA to allow the use of a third booster dose of its Covid-19 vaccine. Food and Drug Administration FDA to allow the use of a third booster dose of its COVID-19 vaccine.
NasdaqMRNA a biotechnology company pioneering messenger RNA mRNA therapeutics and vaccines today announced it has initiated its submission to the US. Currently Pfizer and Moderna are trying to get an authorizationapproval for a booster dose of their respective COVID-19 vaccines. The people spoke on the condition of anonymity before a potential announcement.
Food and Drug Administration FDA for the evaluation of a booster dose of the Moderna COVID-19 vaccine mRNA-1273 at the 50 µg dose level. By Michael Erman and Manojna Maddipatla Reuters Moderna Inc on Wednesday asked the US. The FDA is considering booster shots of the Pfizer Inc and BioNTech SE vaccine but so far has only.
Its not clear when an announcement. Booster campaign after earlier authorization of the Pfizer Inc-BioNTech SE shot. The companies have provided clinical data to support the.
Moderna Inc said on Friday it had asked the EU drugs regulator for conditional approval of a booster shot of its COVID-19 vaccine at a 50 microgram dose. CAMBRIDGE Mass--BUSINESS WIRE--Sep. 1 submitted its application to the US.
Received the Moderna or Pfizer shots or 92 of the total inoculated so far. If the half dose is authorized Moderna could potentially ramp up its global vaccine supply. Food and Drug Administration seeking authorization for a booster shot.
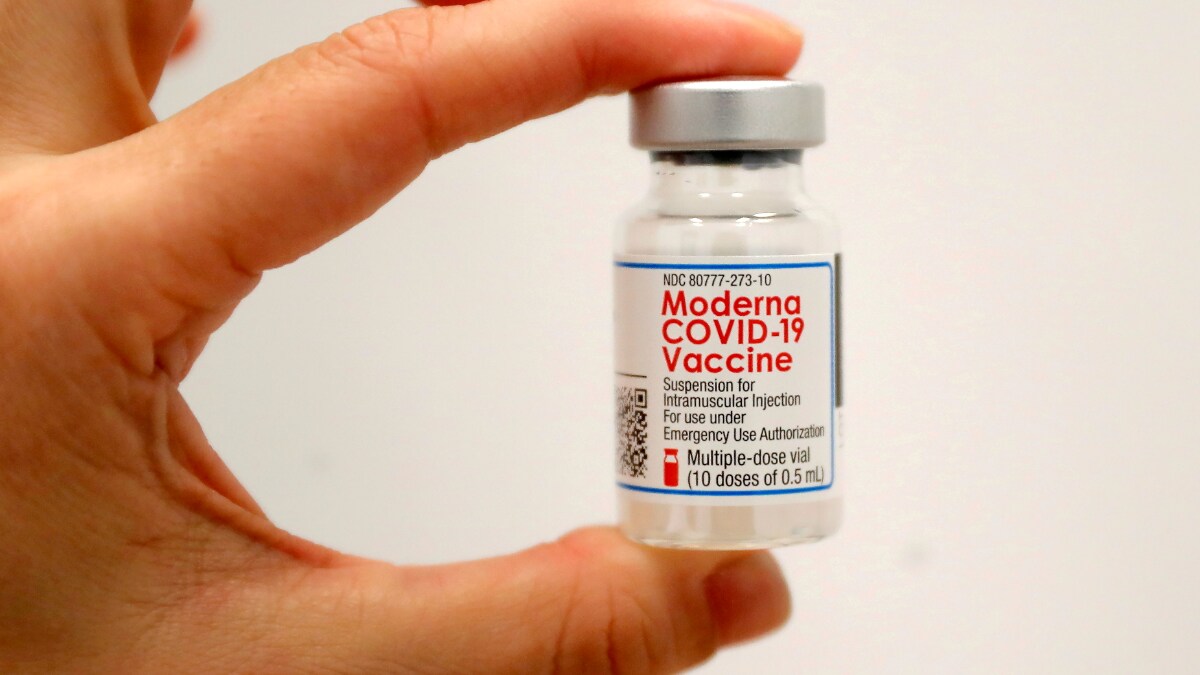
The Moderna vaccine is currently available under emergency use authorization for people age 18 and older in the United States. Moderna Inc on Wednesday asked the US. Data presented by the company indicates that a booster shot containing half the initial dosage is effective at creating a strong immune response.
Boosters will make a bigger financial impact over. Moderna Inc said on Friday it had asked the EU drugs regulator for conditional approval of a booster shot of its COVID-19 vaccine at a 50 microgram dose. The Moderna vaccine is a very similar vaccine to the Pfizer vaccine so I would anticipate an approval for that could follow shortly said Dr.
Moderna COVID-19 Vaccine is authorized for use under an Emergency Use Authorization EUA for active immunization to prevent coronavirus disease 2019. Food and Drug Administration to allow the use of a third booster dose of its Covid-19 vaccine. The original Moderna vaccine.
Moderna Inc said on Friday it had asked the EU drugs regulator for conditional approval of a booster shot of its COVID-19 vaccine at a 50 microgram dose. Moderna Inc on Wednesday asked the US. The company also said it had completed.
Moderna has also filed with the FDA for an emergency use.
Cipla Seeks Govt Help On Pricing Customs Ahead Of Paying 1 Billion Advance For Moderna Vaccine Coronavirus Outbreak News
U S Approves Pfizer Booster Jab Inequality Covid 19 Daily Bulletin Cgtn

Moderna Working On Hybrid Covid 19 Flu Vaccine Wgno Com

Fda Panel Weighs In On Booster Shot Linkedin

Moderna Says Covid 19 Vaccine Booster Will Likely Be Needed Before Winter Wwlp

El Paso County Vaccine Urge Patience After Covid 19 Booster Shots Are Authorized Krdo

Moderna Doses First Subjects In Trial Of Covid 19 Booster Vaccines

Moderna Has Asked The Fda To Authorize A Booster Of Its Covid 19 Vaccine Lakeshore Public Radio
Moderna Says Covid 19 Vaccine Protection Wanes Makes Case For Booster Amnewyork
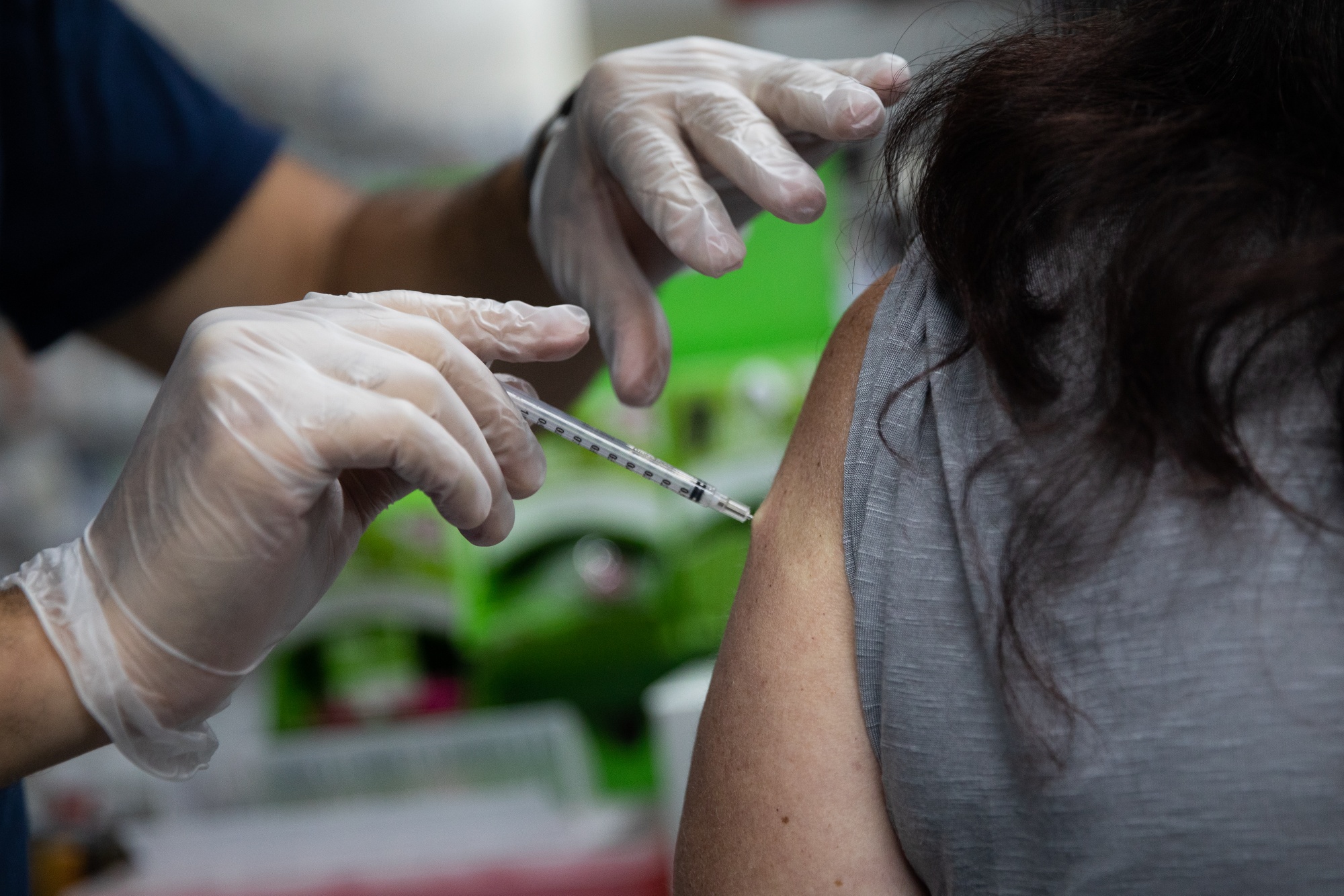
2 Fda Officials Reportedly Resign Over Biden Administration Booster Shot Plan

Moderna Submits Covid Booster To Fda Cbs Boston
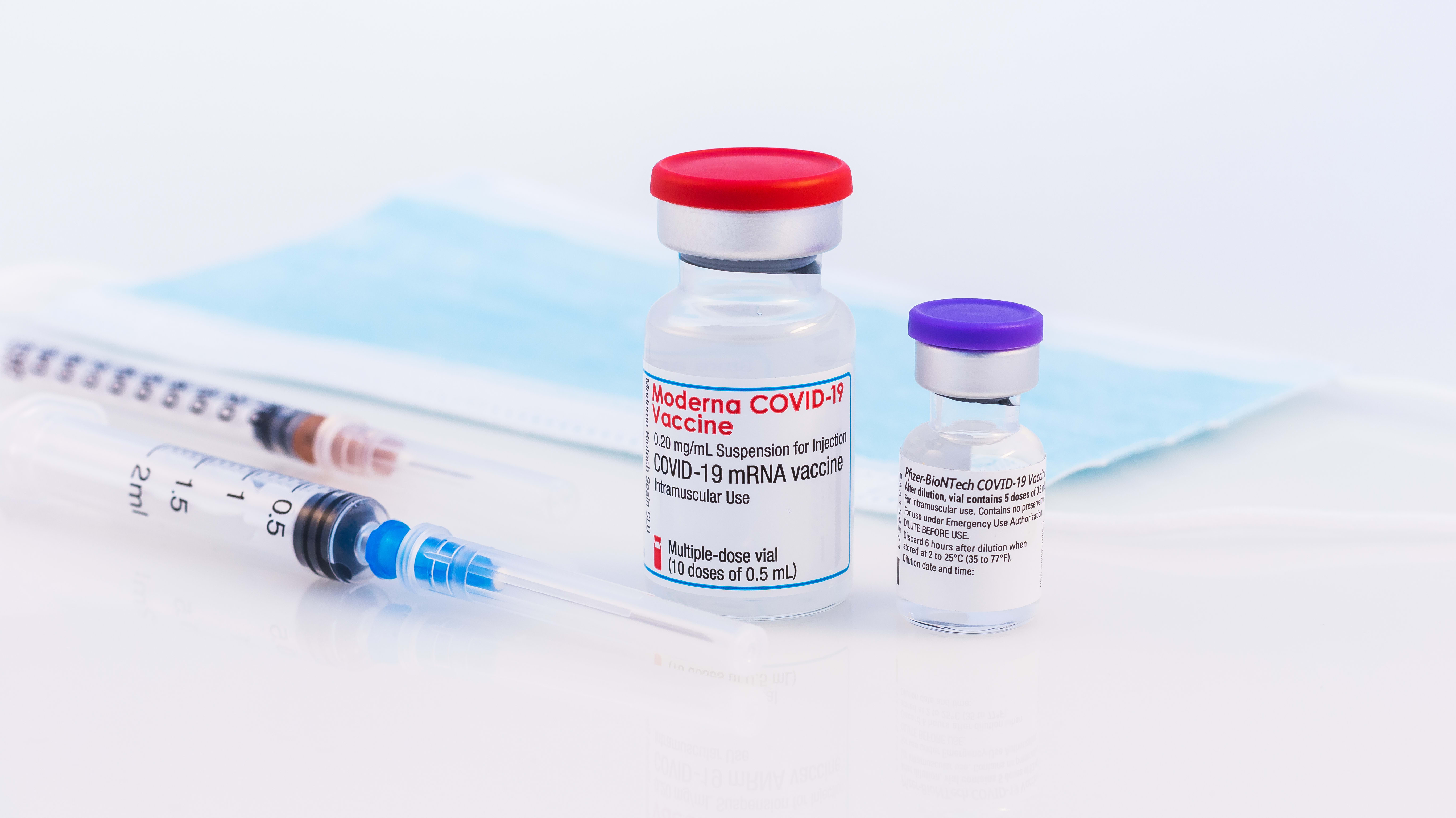
Moderna Seeks U S Authorization For Third Dose Covid Vaccine Booster Bloomberg

Moderna Vaccine Nbc 7 San Diego

Moderna Vaccine Nbc 7 San Diego
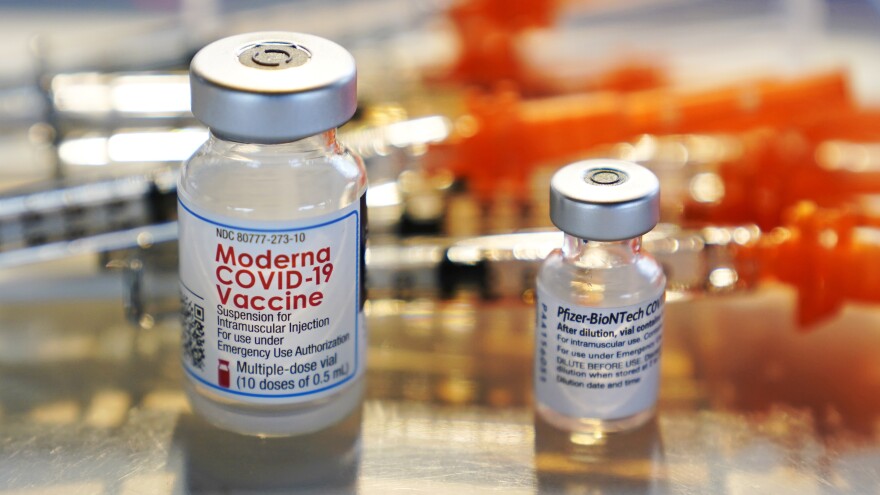
The Biden Administration May Recommend Covid Boosters After 8 Months
:quality(70)/d1hfln2sfez66z.cloudfront.net/09-29-2021/t_4f725635448141f3aeb1f78f3993cc85_name_FDA_may_soon_authorize_Moderna_booster_s_615487d73cae215649f869da_1_Sep_29_2021_15_59_54_poster.jpg)



Post a Comment for "Moderna Booster Authorization"